Overview
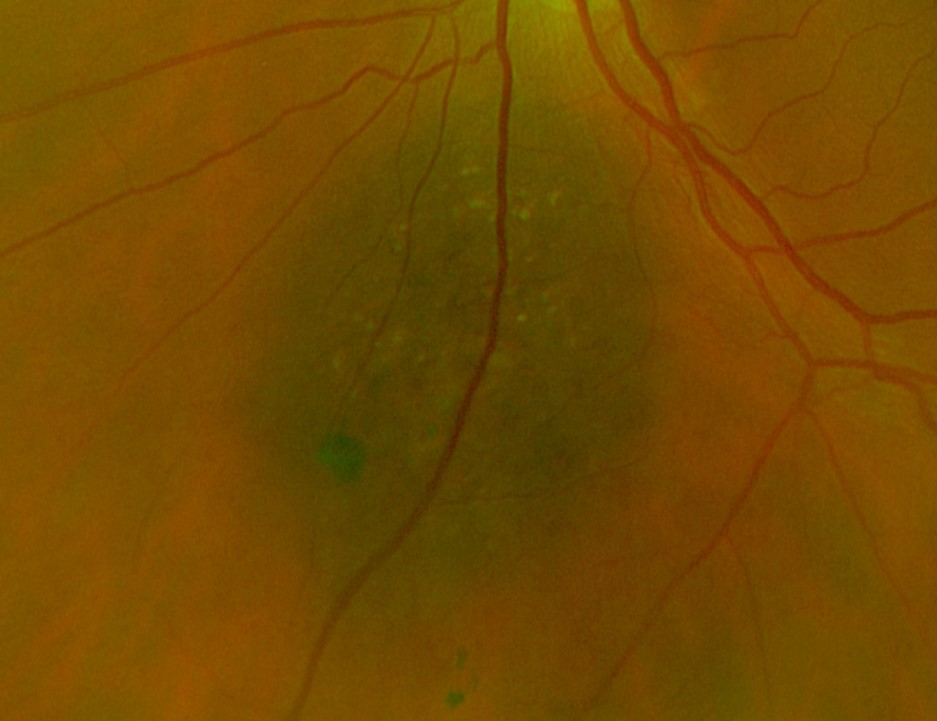
Choroidal naevi are benign melanocytic lesions of the uveal layer.
Clinically, they appears as a round or oval, brown to slate-grey lesion in the fundus typically located posterior to the equator. It is frequently homogenous in appearance but may exhibit pigment mottling or overlying drusen. A green-free filter will enhance its visibility whereas it may become less visible or disappear on red-free imaging.
Naevi may also be amelanotic but mostly show the same imaging characteristics as a melanotic lesion.
Another naevus variant is a halo naevus which appears as a brown-grey choroidal naevus with a surrounding border or halo of depigmentation. The presence of a halo suggests stability of the naevus, however it may be associated with previous cutaneous melanoma (3.3% prevalence compared to 0.3% in a general population (Shields et al., 2010)).
OCT imaging shows hyper-reflectivity within the choroid with posterior shadowing. This can be highlighted with enhanced depth imaging (EDI). Note that this shadowing is absent in an amelanotic naevus. Additional signs include anterior choroidal elevation, overlying drusen, pigment epithelial detachments, atrophy or RPE hyperplasia.
Infrared imaging shows increased reflectivity.
FAF imaging does not show any characteristic autofluorescent pattern, mostly appearing isofluorescent. However, faint hyper-autofluorescence can occur due to the presence of RPE changes such as drusen or pigment epithelial detachments. The presence of overlying lipofuscin (a risk factor for malignancy) will also exhibit hyper-autofluorescence.
Risk of Malignancy
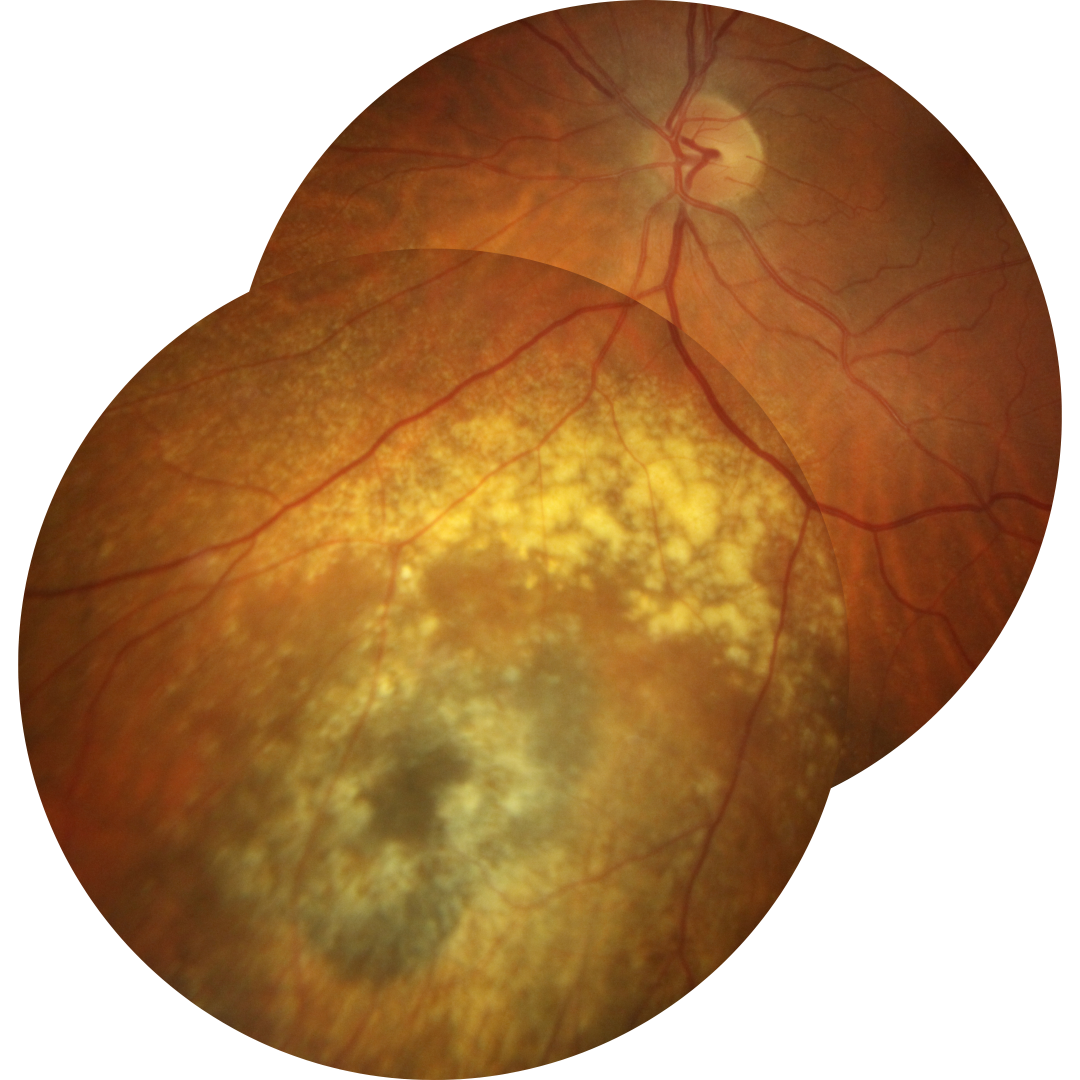
It may be challenging to differentiate between a naevus and a melanoma as the two conditions are on two ends of a disease spectrum.
Several authors have identified approaches to determining the risk of malignancy. The Shields criteria is the most often cited of these and was recently updated (Shields et al. Retina 2019).
"To Find Small Ocular Melanoma Doing IMaging” (TFSOM-DIM):
T = Thickness >2mm
F = Fluid (subretinal fluid present)
S = Symptoms (vision loss)
O = Orange pigment (lipofuscin)
M = Melanoma hollowness (ultrasound biomicroscopy)
DIM= DIaMeter of tumour >5mm
The more risk factors present, the higher the risk of malignant transformation. The 5-year Kaplan–Meier estimation of malignant transformation was found to be 1% with no risk factors present, 11% with 1 risk factor, 22% with 2 risk factors, 34% with 3 risk factors and 4 or more factors give a greater than 50% risk of transformation.
Recently, Oxford Eye Hospital developed the MOLES system to provide guidelines to help distinguish melanoma from naevi. This utilises a scoring system to analyse a variety of clinical features (M=mushroom shape, O=orange pigment, L=large size, E=enlargement, S=subretinal fluid) then providing management recommendations based on this. A 2020 study evaluating these guidelines showed sensitivity of 99.8% for indicating malignancy with a MOLES score of 3 or greater. It also showed that under the guidelines 100% of patients with melanoma would have been referred to an ocular oncologist (Roelofs et al. Cancers 2020).
The full Shields et al paper and MOLES Guidelines may be viewed on the links below.
Case Examples
-
Case 1: Naevus with overlying drusen
An 82 year old asymptomatic Caucasian female with best corrected visual acuity of 6/12 (20/40) in the right eye. The reduced vision is most likely associated with incidental macular changes (a pseudohole and associated epiretinal membrane in the right eye).
Fundus photograph (1) green free (2) and red free (3) images - Right eye
More infoOptomap (1), red separation (2) and green separation (3) images - Right eye
More infoOptomap and Fundus Autofluorescence Imaging (right eye)
More infoSpectralis OCT volume scan taken through the lesion
More infoSpectralis OCT line scans taken through the lesion
More infoB-Scan ultrasound (right eye)
More info -
Case 2: Amelanotic naevus
A 38 year old asymptomatic Caucasian male with best corrected visual acuity of 6/4.8 (20/15) in his right eye.
-
Case 3: Amelanotic naevus
A 45 year old asymptomatic Caucaisan female with best corrected visual acuity of 6/4.8- (20/15-) in the left eye.
-
Case 4: Halo naevus
A 53 year old asymptomatic Caucasian female with best corrected visual acuity of 6/6 (20/20) in the left eye.
Fundus photograph (1), green free (2) and red free (3) images - taken temporal to the left macula
More infoOptomap (1), red separation (2) and green separation (3) images
More infoOptomap and fundus autofluorescence images
More infoSpectralis OCT volume scan through the choroidal lesion
More info -
Case 5: Naevus with overlying drusen
A 63 year old asymptomatic Caucasian female with best corrected visual acuity of 6/6 (20/20) in the right eye.
-
Case 6: Naevus associated with possible choroidal neovascularisation
A 63 year old asymptomatic Asian female with best corrected visual acuity of 6/6 (20/20) in the left eye.
Based on the suspicion of choroidal neovascularisation, this patient was referred to an ophthalmologist for further assessment.
-
Case 7: Suspicious choroidal naevus
A 56 year old asymptomatic Caucasian male with best corrected visual acuity in the left eye of 6/6 (20/20).
Given the presence of exudation and elevation, this patient was referred to an ophthalmologist for further assessment.
-
Case 8: Suspicious choroidal naevus
A 74 year old asymptomatic Caucasian male with best corrected visual acuity of 6/6 (20/20) in the left eye.
Given the presence of subretinal fluid and the proximity of the choroidal naevus to the optic disc, this patient was referred to an ophthalmologist for further assessment.
Fundus photograph (left superior retina)
More infoStereoscopic image of the lesion
More infoOptomap (1), red separation (2) and green separation (3) images - Left eye
More infoSpectralis OCT line scans through the lesion
More infoSpectralis OCT volume scan through the lesion
More infoB-Scan ultrasound (vertical scan)
More info -
Case 9: Suspicious choroidal naevus
A 70 year old Caucasian female complaining of intermittent blurry vision for the past 6 months. Her best corrected visual acuity in the left eye is 6/9 (20/30). Cystoid macular oedema was noted in the left eye.
Based on the clinical appearance detailed below, this patient was referred to an ophthalmologist for further investigations. She was diagnosed with a suspicious choroidal naevus and is being closely reviewed with the ophthalmologist.
-
Case 10: Suspicious naevus showing progression over time
An 82 year old Caucasian female who was referred to an ophthalmologist by the Centre's optometrists in 2011 for investigation of a large suspicious choroidal naevus. The patient currently under the close monitoring of her ophthalmologist.
At her most recent visit she is asymptomatic and visual acuity in this eye is 6/9 (20/30) in the right eye.
Optomap (1), red separation (2) and green separation (3) images - Right eye
More infoOptomap images of the lesion over time
More infoFundus autofluorescence imaging over time
More infoSpectralis OCT volume scans of the lesion in 2020
More infoSpectralis OCT line scans showing change over time
More infoB-Scan ultrasound (right eye)
More info
Differential Diagnosis
References
Bertil Damato, Oxford Eye Hospital guidelines for management of patients with melanocytic choroidal tumours
Jonna et al., 2019. Enhanced depth imaging OCT of ultrasonographically flat choroidal nevi demonstrates 5 distinct patterns. Ophthalmology Retina Volume 3, Issue 3, March 2019, Pages 270-277
Lavinsky, D et al. “Fundus autofluorescence of choroidal nevus and melanoma.” The British journal of ophthalmology vol. 91,10 (2007): 1299-302. doi:10.1136/bjo.2007.116665
Shields CL et al., 2019. Small choroidal melanoma: detection with multimodal imaging and management with plaque radiotherapy or AU-011 nanoparticle therapy.
Shields, CL. et al. (2019) CHOROIDAL NEVUS IMAGING FEATURES IN 3,806 CASES AND RISK FACTORS FOR TRANSFORMATION INTO MELANOMA IN 2,355 CASES, Retina: October 2019 - Volume 39 - Issue 10 - p 1840-1851
Shields CL, et al. Halo Nevus of the Choroid in 150 Patients: The 2010 Henry van Dyke Lecture. Arch Ophthalmol. 2010;128(7):859–864.